Obscurin - Influencing SR architecture and sAnk1.5 protein turnover
The giant obscurin protein is thought to link the contractile apparatus in muscle cells with the sarcoplasmic reticulum (SR), a membrane system specialized in the storage of calcium.
Using the obscurin knockout animal model, we investigated the importance of obscurin for SR architecture and organization. Lack of obscurin in cross-striated muscle cells leads to changes in longitudinal SR architecture and disruption of small ankyrin-1.5 (sAnk1.5) expression and localization. Changes in SR architecture in obscurin knockout mice were also associated with alterations in several SR or SR-associated proteins, such as ankyrin-2 and beta-spectrin. As a consequence, obscurin knockout mice display centralized nuclei in skeletal muscles as a sign of mild myopathy, but have normal sarcomeric structure and preserved muscle function.
|
Model of obscurin functions, and changes to SR architecture in muscles from obscurin knockout mice. Published in the Journal of Cell Science. 2009. Read the full manuscript here > 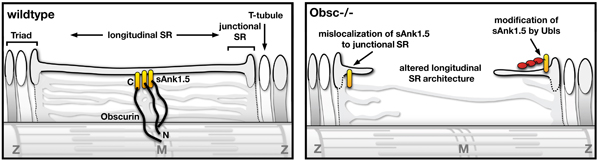 |
In a new manuscript recently published in the journal "Molecular Biology of the Cell", we elucidate the mechanism on how obscurin and KCTD6 regulate the cullin-dependent turnover of small ankyrin 1.5 (sAnk1.5) in heart and skeletal muscle cells. Intriguingly, binding of KCTD6 to sAnk1.5 and its subsequent protein turnover is regulated by posttranslational modifications nedd8, ubiquitin and acetylation of C-terminal lysine residues.
|
Obscurin domain layout and summary of known binding partners. Published in the Journal Molecular Biology of the Cell. 2012. Read the full manuscript here > 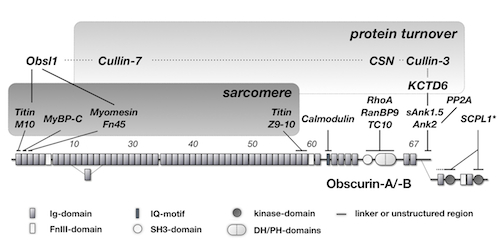 |
Dr. Lange transitioned into a new independent position as Assistant Professor at UC San Diego. He is currently investigating further associations of obscurin and obsl1 to cullin proteins, and cullin-mediated protein turnover.
Section 'Sub' Navigation:
Page 'Breadcrumb' Navigation:

