Research Focus
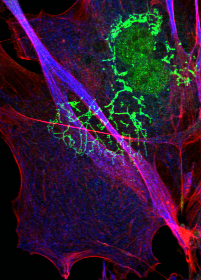
A major focus of the Chen lab is to understand mechanisms by which mutations cause cardiac and skeletal myopathy.
Another major focus of the lab is the study of signaling pathways underlying cardiac hypertrophy and heart failure.
Models utilized in the Chen lab include genetically engineered mouse models and human induced pluripotent stem cells (iPSCs) from human patients. Analyses are performed using physiological measurements, and a range of molecular and cell biological techniques.
Support Basic Research
Support basic research into cardiac and skeletal muscle myopathies. Donate today!
Page 'Breadcrumb' Navigation:
